Olympus Scopes Infection Risk: Why Are Reusable Endoscopes Linked to Serious Outbreaks Across U.S. Hospitals?
In hospitals across the United States, reusable Olympus scopes have been linked to serious infection outbreaks, as well as certain other complications arising from the endoscopic device dislodging or breaking. While these devices play a crucial role in many common diagnostic procedures, their design and use have raised serious health concerns.
Reusable Olympus Scopes: How Does Their Design Create Infection Risks for Patients?
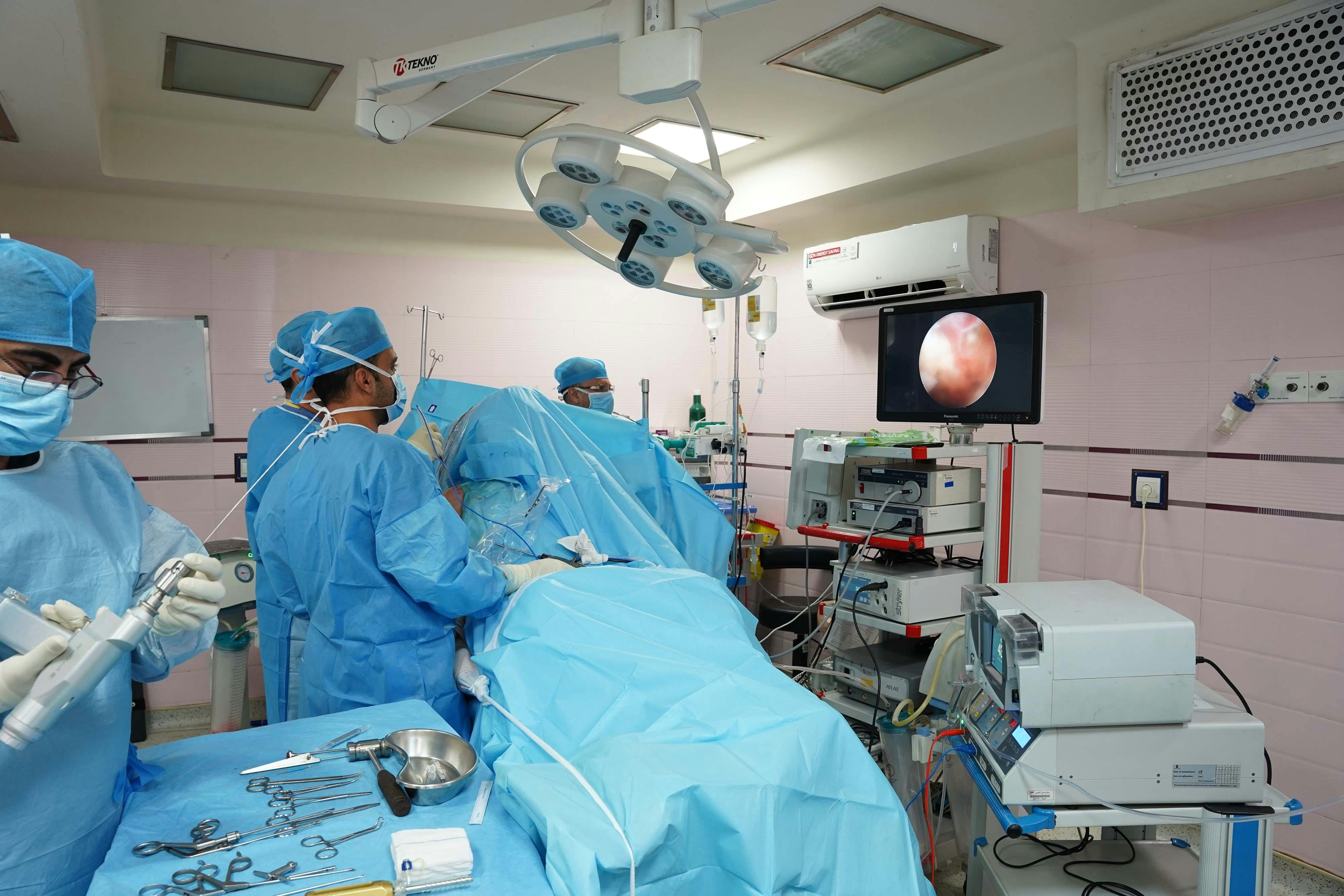
Olympus scopes are specialized endoscopic devices utilized in common gastrointestinal procedures. Each year, these procedures are performed hundreds of thousands of times. But unlike many medical devices, Olympus scopes are reusable and highly complex. The complexity, which includes intricate moving parts and narrow channels, combined with the device being reused for numerous patients, is where the danger lies.
Olympus Scope Sterilization Issues: Why Can’t Hospitals Fully Eliminate Bacteria?
Due to the design of the Olympus scope, it is extremely difficult, if not impossible, to fully sterilize the device. The device includes crevices where biological material can remain trapped even after proper cleaning. Hospitals can strictly follow the manufacturer’s disinfection protocols, and bacteria can still survive. This creates a dangerous situation where a scope used on one patient can unknowingly carry bacteria to numerous subsequent patients.
Similar Post: Did an Olympus Scope Procedure Lead to a Serious Infection? You May Have Legal Options
Undetected Endoscope Infections: Why Are These Outbreaks So Hard to Trace?
As many patients undergo these procedures for unrelated conditions, when a patient later presents with an infection, it may not be immediately linked to the prior endoscope procedure. Therefore, the reusable scopes remain in use, and the underlying infection issue continues to spread to subsequent patients. As Olympus controls a large share of the endoscope market, the potential exposure is extensive and creates a perfect storm of widespread outbreaks. Additionally, many infections linked to these scopes involve drug-resistant organisms which increases the likelihood of severe outcomes. Tuberculosis and HIV infections have also been linked to these devices.
Similar Post: New Olympus Scope Lawsuits: What Patients and Families Need to Know
Medical Device Design Failures: Should Infection Prevention Start Before Devices Reach Hospitals?
The Olympus cases highlight the fact that infection control starts at the device design stage. Failure to take infection prevention into consideration can result in a single contaminated instrument impacting hundreds of patients.
If you believe you were harmed by an Olympus scope, contact Anapol Weiss for a free consultation.
Disclaimer: This blog is intended for informational purposes only and does not establish an attorney-client relationship. It should not be considered as legal advice. For personalized legal assistance, please consult our team directly.