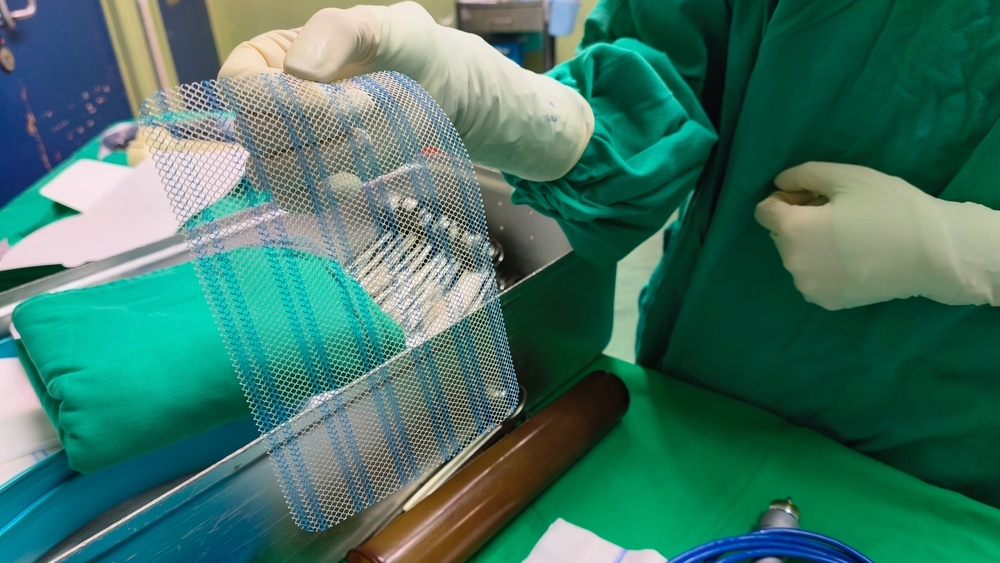
If you had a breast augmentation, a lift, a reduction, or reconstruction after cancer, and something went wrong afterward, there is a chance the problem was not bad luck. There is a chance it was a piece of surgical mesh that never should have been placed inside your body in the first place.
Anapol Weiss is now taking these cases. And if this sounds like your situation, keep reading, because this matters.
Breast Mesh: What Is It And Why Is Everyone Talking About It?
Here is the thing most women were never told. During certain breast surgeries, surgeons started using a mesh scaffold inside the breast to act like a sling, holding the implant in place and keeping the breast lifted. It sounds reasonable enough. The problem is that this mesh was originally made for hernia repairs and pelvic floor procedures. Not for breasts.
Brands like GalaFLEX, Phasix, AlloMax, FlexHD, Strattice, and AlloDerm became widely used in breast surgeries. Surgeons were sold on the idea that it would lead to better, longer-lasting results.
But here is what nobody told patients. The FDA never approved any of these products for use in breast tissue. Not one of them. They were used off-label, meaning no regulatory body ever reviewed whether they were actually safe for this purpose before they went into hundreds of thousands of women.
Breast Mesh Complications: What Happened To The Women Who Got It?
A lot of them got hurt. Badly.
A 2021 clinical review found that more than one in three women who received breast mesh experienced complications. Around 13% needed the mesh surgically removed. A 2024 review found that roughly 10% of patients needed additional surgery just to deal with mesh-related problems, and about 3% lost their implant entirely because of mesh failure.
The complications women reported include chronic infections that antibiotics could not clear, painful hardening of the breast tissue, fluid buildup that kept coming back, the mesh shifting or detaching inside the breast, skin breaking down in ways that sometimes exposed the mesh, nerve damage causing burning or numbness that never went away, and for women who had reconstructive surgery after cancer, complete reconstruction failure.
A lot of these problems did not show up right away. Some women felt fine for months or even years, and then things started going wrong as the mesh degraded or triggered a slow inflammatory response inside the body.
Did Anyone Know This Was Happening: Why Weren’t Patients Told?
Yes. And that is the part that makes this a lawsuit.
Long before lawsuits began, safety concerns about GalaFLEX were raised by Dr. Hooman Noorchashm, a professor affiliated with Northeastern University’s Amy J. Reed Medical Device Safety Collaborative. During a period in which he served as a medical director at BD, he reported that breast cancer recurrence data from clinical trials may not have been fully disclosed to the FDA and that numerous adverse event reports lacked critical details about patient harm.
He went on to file formal petitions with the FDA raising concerns about off-label marketing and the use of these mesh products in breast procedures without proper authorization.
In late 2023, the FDA issued a warning to doctors making clear that no surgical mesh product, synthetic or biologic, is approved for use in breast augmentation, reconstruction, or lifting. Around the same time, GalaFLEX labeling was updated to acknowledge risks tied to off-label breast use.
The concerns were raised. The warnings came late. Women were left dealing with the consequences.
Why Is Anapol Weiss the Right Firm for This: Experience That Changes Outcomes
This is the part where a lot of law firms would give you a list of awards and tell you how great they are. We would rather just tell you what actually matters.
Anapol Weiss has been fighting mesh cases for years. Transvaginal mesh. Y-mesh. Cases that work almost exactly the same way breast mesh cases work. We know what documents to go after. We know what arguments manufacturers will make. We know how to counter them. You are not hiring a firm that is learning on the job here.
Our firm has recovered more than 1.4 billion dollars for people harmed by defective medical devices. That number matters not because it is impressive but because it means manufacturers know who we are and what happens when they refuse to settle fairly.
Anapol Weiss also runs a dedicated Women's Health Litigation practice. That is not a branding exercise. It reflects the reality that some of the most harmful defective medical devices of the last 20 years were placed inside women's bodies. Pelvic mesh. Breast mesh. Hormonal devices. We have been litigating these cases for a long time and we understand what these injuries actually mean for the people living with them.
On the practical side, breast mesh cases are likely to consolidate into a Multi-District Litigation proceeding. That is a coordinated national lawsuit involving thousands of plaintiffs. Firms that file early and have MDL experience tend to get leadership roles that directly shape how settlements are structured. Anapol Weiss has offices in Philadelphia, Dallas, Los Angeles, Minneapolis, Washington D.C., New Jersey, and Arizona. We operate at that level.
And you will not pay anything upfront. Every breast mesh case we take is on contingency. We cover all the costs. We only get paid if we win money for you. If we do not win, you owe us nothing at all.
Who Can File a Breast Mesh Lawsuit: Do You Qualify?
You may qualify if you had any of the following and later developed complications:
- Breast augmentation using a mesh scaffold
- Breast lift using an internal bra technique
- Post-mastectomy reconstruction with AlloDerm, Strattice, GalaFLEX, FlexHD, Phasix, AlloMax, or similar products
And the complications that tend to support the strongest cases include infections that required surgery, fluid buildup requiring drainage or reoperation, the mesh shifting or causing the implant to drop, tissue damage or necrosis, implant loss, chronic pain or nerve issues, or any revision surgery related to the mesh.
If you are not sure whether mesh was used in your surgery, your operative report will say. That is the document from the day of surgery that lists every material implanted. We can help you get it.
Breast Mesh Lawsuit FAQs: What Women Are Asking Right Now
My surgery was years ago. Is it too late?
Maybe not. Many states use what is called a discovery rule, which means the clock on your deadline to file starts when you knew or reasonably should have known your injury was connected to the mesh. Since complications often appear long after surgery, a lot of women who had procedures years ago are still within the window. The only way to know for sure is to talk to us. Do not assume you are out of options before you check.
I had reconstruction after breast cancer, not elective surgery. Does that matter?
It matters in a good way. Post-mastectomy reconstruction cases are some of the most powerful in this litigation. Products like AlloDerm and Strattice were heavily marketed to reconstructive surgeons without FDA clearance for that purpose. Women who endured cancer treatment and then had their reconstruction fail because of mesh have compelling claims, and those stories resonate deeply with juries.
My surgeon was the one who recommended the mesh. Can I still file?
Yes. These lawsuits go after the manufacturers, not your surgeon. The core of the case is that manufacturers sold these products for breast use without approval and without properly warning about the risks. Your surgeon may have genuinely believed the product was safe based on what manufacturers told them. That does not let the manufacturer off the hook.
What complications actually qualify?
Anything serious that required medical intervention. Infection, fluid buildup, hardening of the breast, the mesh moving or detaching, implant loss, skin breakdown, chronic pain, nerve issues, revision surgery. If you had to go back into surgery because something went wrong with the mesh, that is exactly the kind of case we want to hear about.
I signed a consent form before surgery. Does that mean I gave up my rights?
No. Consent forms cover general surgical risks. They do not release manufacturers from their own separate legal duty to warn about known product risks. If the company that made your mesh failed to disclose that it had never been approved for breast use and that serious complications were occurring, your signature on a pre-surgery consent form does not protect them.
Do I have to go to court?
Almost certainly not in person. The vast majority of mass tort cases resolve through settlements, not individual trials. In coordinated litigation, a small number of cases go to trial to help set the value of settlements for everyone else. Most plaintiffs never appear in a courtroom. That said, Anapol Weiss will go to trial if manufacturers do not offer fair compensation. That willingness is part of why they settle.
What will this cost me?
Nothing upfront. Nothing at all unless we win. We cover medical record retrieval, expert witnesses, filing fees, and every other litigation cost. Our fee comes out of the recovery if and when we get one for you. There is no financial risk to calling us.
What records do I need to gather?
The most important thing is your operative report from the surgery, which identifies the mesh product. Beyond that, any medical records showing complications, treatment, additional surgeries, or ongoing symptoms. If you do not have these yet, do not worry. Our team has a lot of experience tracking them down. Just start by calling us and we will walk you through it.
Talk to Us. It Costs You Nothing.
If any of this sounds familiar, please reach out. You do not need to have all the answers or all the paperwork. You just need to tell us what happened.
Call Anapol Weiss at 215-735-1130or submit our online contact form to schedule a free, confidential consultation. No pressure, no fees, no obligation. We only get paid if we win your case.
Anapol Weiss | Nationally Recognized Trial Attorneys | Philadelphia, PA | This blog is intended for informational purposes only and does not establish an attorney-client relationship. It should not be considered as legal advice. For personalized legal assistance, please consult our team directly.